Chem. Mater. 30 (2018) 7183–7200

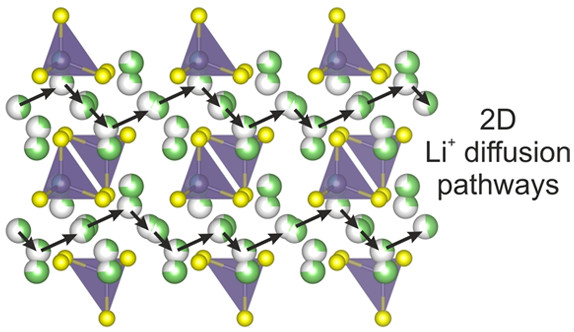
To understand the structural and compositional factors controlling lithium transport in sulfides, we explored the Li5AlS4-Li4GeS4 phase field for new materials. Both parent compounds are defined structurally by a hexagonal close packed sulfide lattice, where distinct arrangements of tetrahedral metal sites give Li5AlS4 a layered structure and Li4GeS4 a three-dimensional structure related to γ-Li3PO4. The combination of the two distinct structural motifs is expected to lead to new structural chemistry. We identified the new crystalline phase Li4.4Al0.4Ge0.6S4, and investigated the structure and Li+ ion dynamics of the family of structurally related materials Li4.4M0.4M'0.6S4 (M = Al3+, Ga3+ and M' = Ge4+, Sn4+). We used neutron diffraction to solve the full structures of the Al-homologues, which adopt a layered close-packed structure with a new arrangement of tetrahedral (M/M') sites and a novel combination of ordered and disordered lithium vacancies. AC impedance spectroscopy revealed lithium conductivities in the range of 3(2) × 10-6 to 4.3(3) × 10-5 S cm-1 at room temperature with activation energies between 0.43(1) and 0.38(1) eV. Electrochemical performance was tested in a plating and stripping experiment against Li metal electrodes and showed good stability of the Li4.4Al0.4Ge0.6S4 phase over 200 h. A combination of variable temperature 7Li solid state nuclear magnetic resonance spectroscopy and ab initio molecular dynamics calculations on selected phases showed that two-dimensional diffusion with a low energy barrier of 0.17 eV is responsible for long-range lithium transport, with diffusion pathways mediated by the disordered vacancies while the ordered vacancies do not contribute to the conductivity. This new structural family of sulfide Li+ ion conductors offers insight into the role of disordered vacancies on Li+ ion conductivity mechanisms in hexagonally close packed sulfides that can inform future materials design.
The American Chemical Society holds the copyright for the published article. More information can be found on their website.
Videos of the ab initio molecular dynamics trajectories presented in the paper are available below.