Journal of the American Chemical Society, 133 (2011) 1016–1032

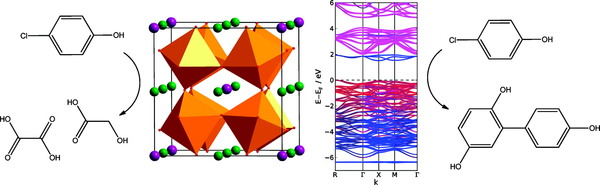
Charge transfer between metal ions occupying distinct crystallographic sublattices in an ordered material is a strategy to confer visible light absorption on complex oxides to generate potentially catalytically active electron and hole charge carriers. CaCu3Ti4O12 has distinct octahedral Ti4+ and square planar Cu2+ sites and is thus a candidate material for this approach. The sol-gel synthesis of high surface area CaCu3Ti4O12 and investigation of its optical absorption and photocatalytic reactivity with model pollutants are reported. Two gaps of 2.21 and 1.39 eV are observed in the visible region. These absorptions are explained by LSDA+U electronic structure calculations, including electron correlation on the Cu sites, as arising from transitions from a Cu-hybridized O 2p-derived valence band to localized empty states on Cu (attributed to the isolation of CuO4 units within the structure of CaCu3Ti4O12) and to a Ti-based conduction band. The resulting charge carriers produce selective visible light photodegradation of 4-chlorophenol (monitored by mass spectrometry) by Pt-loaded CaCu3Ti4O12 which is attributed to the chemical nature of the photogenerated charge carriers and has a quantum yield comparable with commercial visible light photocatalysts.
The American Chemical Society holds the copyright for the published article. More information can be found on their website.